 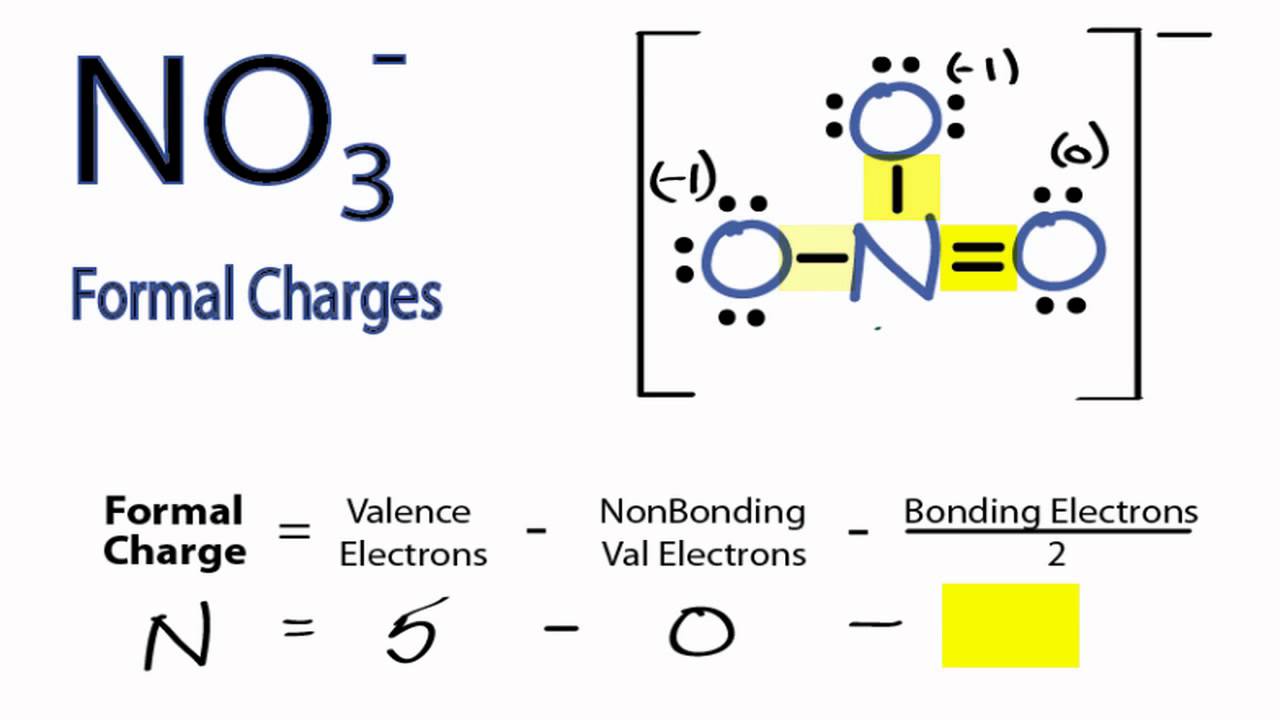 
The difference between the atom's number of valence electrons and the number it owns is the formal charge. Count all of its lone pair electrons, and half of its bonding electrons. Since sulfur has six valence electrons, we conclude that two electrons are not involved in the bonding, i.e., that there is a lone pair.\) have the same strength and length, and are identical in all other properties. To find formal charges in a Lewis structure, for each atom, you should count how many electrons it 'owns'. Remember, electron counting to determine an octet counts all of the bonding and nonbonding electrons equally. 
If it is two short, it has a two plus charge. Formal Charge (number of valence electrons in neutral atom)- (non-bonded electrons + number of bonds) Example 1: Take the compound BH4 or tetrahydrdoborate. Without experimental data, calculating of the formal charge on each atom. 
The difference between the number of valence electrons present in an atom. the formal charge is additive: if the atom has two extra electrons in the molecule, it has a two minus charge. Experimental evidence, such as bond length, is the best verification of structures. In the sulfite ion, SO 3 2– for example, the oxidation number of sulfur is +4, suggesting that only four sulfur electrons are involved in the bonding. In this video we cover How to calculate formal charge faster Watch this video to understand How to calculate formal charge faster without the complicated fo. or q) in the covalent view of bonding is the charge assigned to it by the covalent view of bonding, which assumes that electrons in all chemical bonds are shared equally across atoms, regardless of relative electronegativity between them. Oxidation numbers can sometimes also be useful in writing Lewis structures, particularly for oxyanions. This type of reaction can be recognized because it involves a change in oxidation number of at least one element. Wikipedia A formal charge (FC) is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity. Lewis structures practice work, Chemistry 11 ap formal charge vsepr work, Lewis structure work. Oxidation numbers are mainly used by chemists to identify and handle a type of chemical reaction called a redox reaction, or an oxidation-reduction reaction. practice worksheet to solve practice problems on calculating formal charges for atoms in the strictures given in the practice problems. Calculate the formal charge on each atom in the molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
